Why Publication Extenders Need To Be Integrated in Your Publication Planning From the Start
Publication extenders are an indispensable part of any well-rounded publications strategy — including those designed to support a new drug launch. Yet when it comes to mapping out development and dissemination, these short-form content assets are still all too often treated as an afterthought.
That’s a problem. After all, publication extenders play a key role in helping time-crunched healthcare professionals (HCPs) engage with the latest scientific data related to the new therapy you’re launching. But you can’t get the most value out of extenders if they aren’t carefully integrated into your publication strategy and plan.
Let’s take a closer look at why you should include extenders from the earliest stages of your publications strategy development — and how to effectively incorporate extenders into your publication plan.
What Are Publication Extenders, and What Role Should They Play in Your Drug Launch Strategy?
Publication extenders are microcontent pieces featuring a publication’s high-level takeaways in the bite-sized formats HCPs prefer. For instance, an extender may take the form of an infographic, a short explainer video or a downloadable one-sheet featuring a bulleted list of key data.
Extenders are crucially important because they meet today’s HCPs where they are — that is, facing limited time to devote to continuing education and struggling to keep current with an overwhelming torrent of new literature.
HCPs still view journal publications as the premiere source of trustworthy scientific research. But they also overwhelmingly prefer to consume that content in interactive, bite-sized pieces they can actually digest.1
That’s where publication extenders come in. They intentionally bridge the gap between your full-length publications and the harried physicians you most want to engage and educate. In the context of a drug launch, extenders are foundational in making sure the HCPs and KOLs in your field are aware of and engaged with the most up-to-date and relevant scientific data as they evolve.
The Ideal Time to Work Publication Extenders Into Your Publication Plan
As a medical communications professional, you’re already used to planning publications on an annual cadence. But assuming you’re one of the more modern organizations that creates publication extenders, there’s a good chance you’ve been treating them differently. You’ve likely been in the habit of ideating and developing those assets only after each publication is released.
This lagging planning cycle for extenders might seem logical given the perennial uncertainties you’re so used to dealing with. For instance, during the annual planning period it can be difficult to pinpoint which manuscripts should take priority to support the therapeutic area objectives. In addition, you may lack clarity about how to allocate the necessary budget to create extenders; by waiting, you can make decisions based on the actual remaining budget. On top of that, shifting timelines inevitably inject uncertainty in the planning process, and publication extenders only add to the complexity.
Despite these valid concerns, the ideal time to map out the specific publication extenders you’ll create to support your drug launch strategy in the coming year is during your annual publication strategy and planning process.
4 Benefits of Pre-Planning Publication Extenders
Publications — and therefore publication extenders — play an important role in every step of the drug launch process, including the early market, pre-launch, launch and post-launch phases. Including extenders in your publication planning as early as possible allows you to make the most of these assets at every step along the way.
But early planning yields a range of other benefits, too. These include:
1. A more robust, strategic portfolio of publication extenders
When you ideate and develop publication extenders after the fact, you’re more likely to go with assets that are simple and easy to produce. You might even find yourself dismissing formats that require greater planning and longer lead times, even when those formats are more strategically desirable.
Planning publication extenders for the upcoming year allows you to take a much more strategic approach. It gives you the opportunity to leverage recent insights and take a holistic view of therapeutic area priorities and audience needs. You also give yourself the chance to intentionally align your publication extenders with the milestones you hope to achieve as you progress your launch strategy over the year.
Finally, planning ahead enables you to produce a higher volume of extenders with a wider variety of formats over the course of the year. And that equals increased awareness and HCP engagement.
With these advantages, you can more easily identify topics and formats based not on expedience, but on their strategic value. As a result, you’ll develop a series of publication extenders that efficiently convey key communication points to your audience, maximize the impact of publications throughout the year — and support your overarching launch strategy.
2. A smoother content production process
If you scramble to create extenders after the ink dries on a new publication, you may find the authors involved in the full-length manuscript are no longer available to assist with related extenders.
Planning ahead allows you to appropriately set expectations with the agency partners and experts you want to include in the extender creation process. Letting authors know they’ll be involved not only in developing the manuscript but also in the creation of, say, a podcast and an author video, tees them up to make time and space to collaborate — and approach those projects thoughtfully.
Beyond that, pre-planning extenders enables you to properly anticipate the resources needed to bring those materials to fruition, including writing, editing and design experts.
3. Greater internal alignment
Pre-planning publication extenders gives you the chance to liaise with the broader medical affairs team and other cross-functional departments, creating greater alignment on priority objectives. This internal clarity can foster synergies to collectively achieve company goals as you move toward a new drug launch.
It also provides an opportunity to establish connections between the activities planned by other medical affairs functions (including satellite symposiums, congress materials and MSL materials). This allows cross-functional teams to work cohesively toward common goals, solidify priorities and provide a clear vision for the year.
For instance, you might find that a particular set of extenders can be used both on a company-owned HCP engagement platform and in a scientific deck used by MSLs to interact with HCPs. Identifying this crossover potential early on eases content planning for your HCP engagement platform and aids discussions with Field Medical leadership in an earlier stage.
It’s worth noting that this move toward increased content planning alignment is in keeping with a broader trend in the medical affairs industry: the increasing integration of various functions to collectively pursue common objectives.
4. Efficiency gains and cost reductions
Time and budget limitations are the single biggest hurdle when it comes to developing publication extenders.2
The good news? Having a clear, overarching plan that spells out which extenders you’ll develop over the course of each year creates serious efficiency gains and cost reductions.
Having visibility into the various extender formats allows you and your agency partner to develop extenders using a modular content approach. You can then create a library of modules (i.e., written content and visuals) that can be repurposed across various extenders.
How to Include Publication Extenders in Your Annual Publication Plan
Traditional publication plans focus primarily on full-length journal publications (including abstracts and summaries) and related materials for congresses. Innovative publication plans, on the other hand, give publication extenders the same treatment and representation.
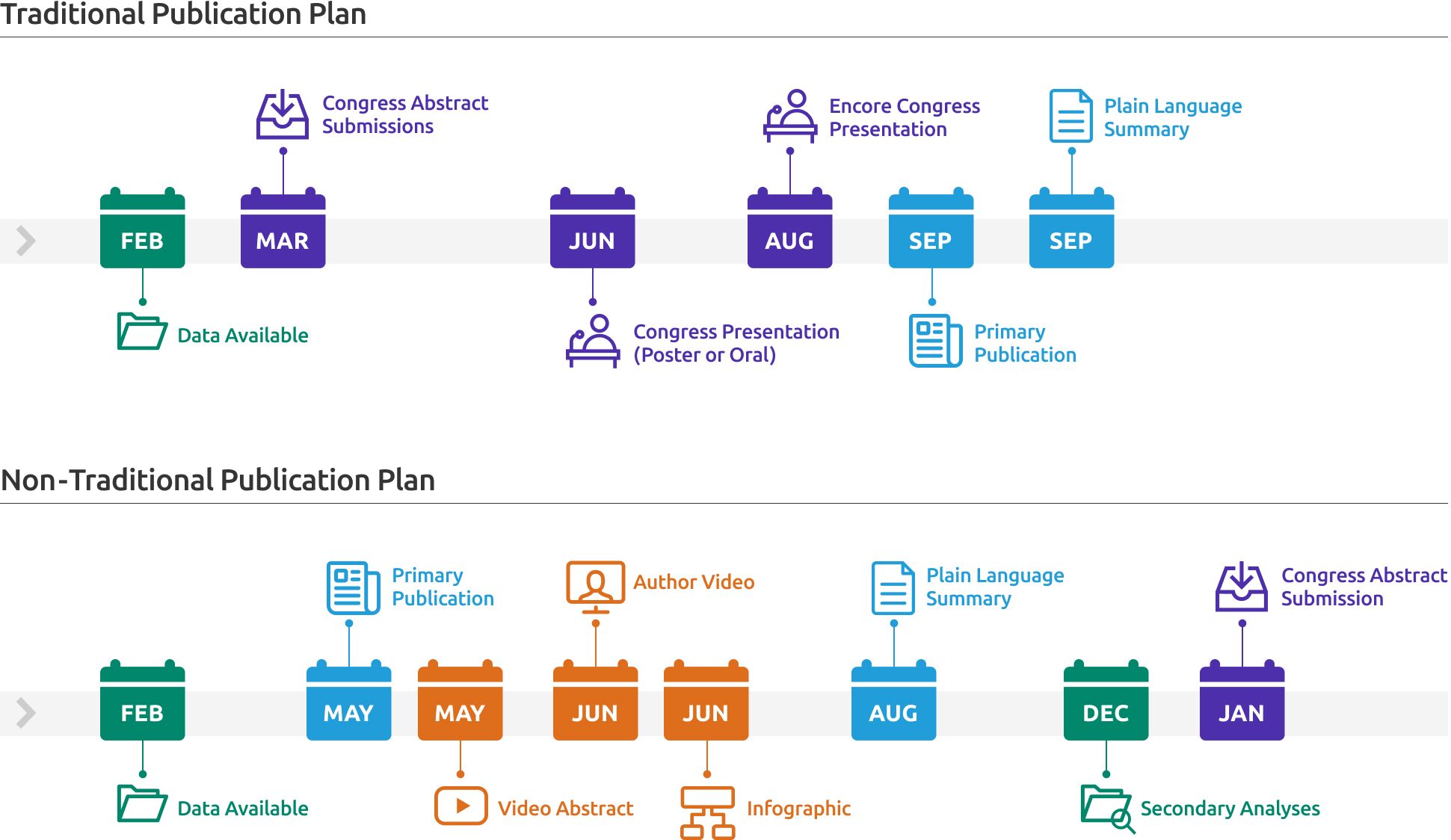
Note. Whann, B. & Rozich R.A. (2023). Beyond the Traditional Publication Plan. ISMPP Academy. Philadelphia, PA, USA
The first step is to identify which publications merit publication extender treatment, as well as which formats to develop. A systematic approach is best. To that end, a publication selection matrix guides your publication team in choosing and prioritizing key publications, making it simpler to plan extenders accordingly.
Finally, consider collaborating with an external medical communications agency like MedComms Experts to develop your drug launch communications plan. A strong strategic partner can:
- Assist in prioritizing and allocating your budget where it makes the most impact.
- Advise on the channel and tactics to leverage to deliver the extenders.
- Set metrics to track the impact of your publication extender strategy.
- Summarize key initiatives and associated metrics concisely for presentation to leadership, thereby enhancing the value of your publication extender strategy within the broader organization.
Ready to take your publication extender portfolio to the next level and increase the impact of your drug launch strategy? Let’s talk.
